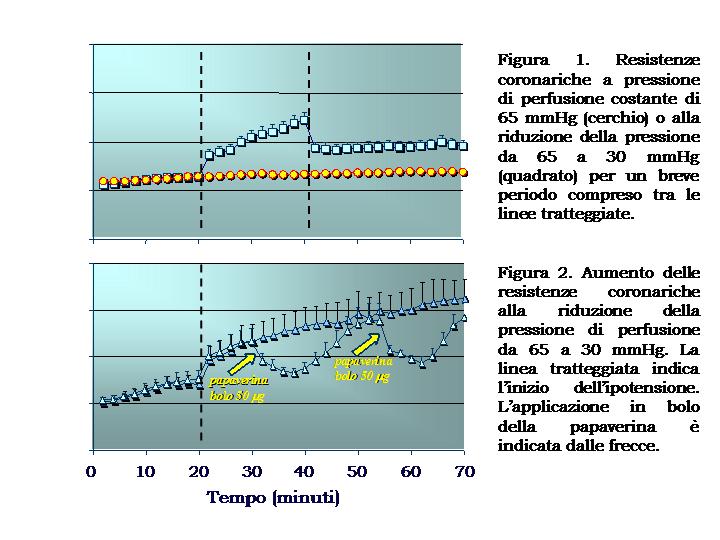
According to the classical model of cardiovascular physiology, the control of coronary vascular tone is tuned to guarantee the continuous adjustment of flow to tissue requirements (metabolic control) and its independence from variations in aortic pressure (autoregulation or myogenic control). However, this model does not encompass the control of capillary pressure within optimal values of Starling equilibria and even obstacles it to some extent. During ischemia, in particular, this model assumes the existence of a maximal vasodilation and a low intravascular pressure. This theory hardly challenges the common assumptions of vascular physiology and, mostly, does not account for recent data (also obtained in our Institute) documenting that both primary and secondary ischemia were associated with a severe microcirculatory vasoconstriction, in patients with coronary artery disease. Nevertheless, its active vasoconstrictor nature was documented by the superselective administration of adenosine. In fact, this substance induced a marked vasodilation in the ischemic region and improved the electrical signs of ischemia without any change in systemic hemodynamics. Altogether, these findings bring us to hypothesize that microvascular constriction plays some role in the precipitation and maintenance of ischemia in patients with coronary artery disease. Several lines of evidence now corroborate this hypotesis, however, the molecular mechanisms triggering and maintaining this reaction are not fully understood. The paradoxical vasoconstrictive response, even if observed in patients affected by ischemic cardiomyopathy, could not be a mere pathological phenomenon. In fact, it might represent a physiological mechanism working for the control of capillary pressure by means of an opposite mechanism than myogenic reflex and with a regional rather than a global effect. Recently, we developed an experimental model of isolated mouse heart, studied in Langendorff configuration to evaluate the response of microvascular tone to changes in perfusion pressure. In a study involving more than 150 mouse hearts we documented that a reduction in perfusion pressure caused a biphasic increase in coronary resistance with a steep slope in the first ten minutes followed by a slower progression. This increase was reversed by bolus injection of papaverine, as one would expect from the operation of an active process. Pharmacological experiments pointed out that the vasoconstrictor response to hypotension requires the integrity of both NO and ET-1 (by means of ETa receptors) systems and the likely contribution of ROS being generated by the reduction of flow.
In summary, our study indicated that the vasoconstrictor response to hypotension represents a physiological adjustment of coronary microcirculation in which the two endothelial mediators (nitric oxide and endothelin) act synergically, even though in the great majority of models reported in literature they are opposite in action.
Immagini: